Bleaching is an important and essential step in pre-treatment of textiles. It helps ‘whiten’ the textile material by removing undesired inherent colouring components. Generally, the natural fibres and their blends comprise about 50 per cent of total textile substrates and are invariably bleached, while the regenerated and synthetic goods are given this treatment considering the specific requirement. Depending on the substrate, a number of chemicals and application methods are employed to achieve optimum bleaching effect, however, it is also imperative to remove and get rid of the excess and leftover bleaching agent from the textile goods before subsequent wet processing. This article is an attempt to review various bleaching chemicals used in the textile industry and the methods of their removal and cleaning.
Inherent Colourants in Textile
Natural vegetable fibres like cotton, linen, jute etc contain varying extent of pigments like chlorophyll, xanthophyll and carotene. This imparts off-white, yellowish-brown hue and depends on the area of cultivation, climatic conditions and varies from crop to crop. Similarly, animal fibres like wool and silk too contain non-protein impurities which impart characteristic colour.
Need of Bleaching
Bleaching is the process of decolourisation of raw textile material by removing inherent and or acquired colouring components from the fibre. It provides base whiteness to the textile material which could be further whitened with the help of optical brighteners or dyed/printed depending on the desired end use. Even in case of regenerated and synthetic fibres, bleaching step is incorporated for achieving full white or extra bright shades. However, bleaching step could be omitted in case of material to be dyed with dark and dull shades.
Bleaching can be carried out at various stages of conversion of fibre to garment i.e. from fibre, yarn, hank, woven, knit, towel, sewn up garments etc on various types of machines by simple hand processing to sophisticated bleaching ranges and by different application processes from exhaust to continuous.
Bleaching Chemicals
Though bleaching is an age-old process, the major commercial chemicals were developed in the last few centuries. The modern bleaches resulted from the work of 18th century scientist Claude Berthollet, who developed bleaching with sodium hypochlorite, and later Louis Thenard produced hydrogen peroxide, which gained wide commercial acceptance.
Basically, two types of chemical processes are employed – oxidative and reductive – and are employed depending on the substrate to be bleached and the nature of inherent colouring impurities.
The organic compounds containing conjugated double bonds are considered to be the colour producing agents in natural fibres. Their decolouration can occur by breaking up the chromophore, most likely by destroying one or more of the double bonds within the conjugated system and by breaking up the resonance.
• An oxidative bleach works by breaking the chemical bonds that make up the chromophore and changes the molecule into a different substance that does not absorb visible light.
• A reductive bleaching works by converting double bonds in the chromophore into single bonds by the addition of one mole of hydrogen which eliminates its ability to absorb visible light.
In case of oxidative bleaching, chlorine is the basis for commonly used bleaching agents like sodium hypochlorite, bleaching powder and sodium chlorite. On the other hand, the non-chlorine containing oxidative chemicals are hydrogen peroxide, sodium persulphate, sodium percarbonate, sodium perborate and peracetic acid. At times peroxide bleaches are also used along with catalysts and activators like tetra acetyl ethylene diamine (TAED) and sodium nonanoyloxy benzene sulfonate (SNOBS). The reducing agent based bleaching agents comprise of sodium dithionite or sodium hydrosulphite, sodium thiosulphate and sulphinic acid derivatives.
The commonly used application methods are:
• Batch or discontinuous - Kier, Jigger, Winch, package, softflow, jet
• Semi continuous – pad batch or pad roll
• Continuous - J–box, pad steam
The oxidative chemicals are widely used for bleaching of natural fibres like cotton, linen, jute, wool, while reductive method is used for polyamide and polyacrylates. The chlorine-based bleaches, though economical and easy to apply, have lost importance due to health and environmental issues, but they still find use in some areas.
The selection of bleaching agent depends on the fibre and machinery in use, the method of application, the extent of whiteness expected without adversely impacting fibre strength and meeting the desired cost economics.
Hydrogen peroxide is the most common and universally used bleaching agent and is applied under specified conditions of time, temp, pH and stabilisers for optimum efficacy. The generally considered advantages in its use are:
• Less weight loss than that with hypochlorite bleaching
• Bleached goods are more absorbent than hypochlorite bleached goods
• Less yellowing of post bleached white goods
• Suitable for bleaching of cotton, wool and silk
• Suitable for combined and continuous scouring and bleaching
Need for Bleach Clean-up
Generally, the peroxide bleaching process is set in such a way that it retains about 10-15 per cent of the initial peroxide amount at the end. This is achieved by optimising the process parameters and incorporating suitable stabilisers. The basic purpose is to avoid uncontrolled and rapid decomposition of strong oxidising agent which is detrimental to the tensile strength of cellulosic textiles. Similarly, in case of other bleaching agents, in order to achieve optimum results, slight excess is used at the start which is carried forward as residue at the end.
When the textile material is to be dyed after bleaching, it becomes imperative that the left over/residual bleaching agent is effectively removed, as the bleaching agent affects colouring component/chromophore of dyestuff. Failure to do so can result in poor dyeability, batch-to-batch shade reproducibility as well as uneven dyeing. Therefore, in order to get rid of the remaining leftover bleaching agent, an intermittent bleach clean-up process is incorporated before dyeing. It is also termed as peroxide killing or neutralisation in case of hydrogen peroxide and antichlor in case of chlorine-based bleaching. This is specifically required in processes involving use of material in package or rope form, while in continuous process, owing to the open width form and continuous counter current water washing, generally bleach clean-up is not required.
Even in case of full bleach or full white process, where high concentration of bleaching agent is used, it is considered advantageous to neutralise and clean-up to avoid subsequent yellowing during extended storage conditions.
The Direct and Reactive dyestuffs are generally sensitive to oxidising agents and even small amount of post bleaching residual hydrogen peroxide may have a negative influence on subsequent dyeing resulting in loss of depth. The Vat and Sulphur dyes are relatively stable and unaffected by the residual bleaching chemical. However, it is still considered advisable to clean-up the strong oxidising agent before the start of dyeing in order to avoid neutralisation of dye solubilising chemicals which could cause inadequate reduction/precipitation of this class of dyestuffs.
Given below is an illustration on dyeability and effect on colour strength of select high exhaust bis-monochlorotriasine type Reactive dyes – with and without bleach clean-up.
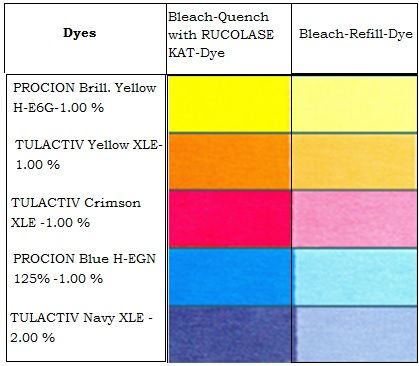
Methods of Beach Clean-up
Various methods of bleach clean-up are employed in the textile industry. These involve repetitive washing, catalytic decomposition, reductive neutralisation and enzymatic killing.
1. Washing with fresh water: This was the popular method employed in the past and still adopted in some areas for complete removal/depletion of the left-over oxidising agent. Though this is a simple process, it
• requires high amount of water for repeated washing
• increases energy consumption for heating water which is unprofitable
• lengthens processing time due to draining and refilling of the machine
2. Catalytic decomposition: This involves the use of salts of selected heavy metal ions which, under controlled temp and alkaline pH conditions, rapidly activate rate of decomposition of residual hydrogen peroxide and thereby destroying it completely. Though the process is simple, fast and cost-effective, the disadvantage is if the residual peroxide content is high and the process is not controlled adequately, it may result in degradation of cellulose causing breakages in yarn and pin holes in fabric.
3. Reductive neutralisation: It is based on use of inorganic salt based reducing agent like RUCORIT INPK which works at the equal concentration of the residual peroxide and effectively cleans it up. The specific advantages of this product are:
• cost economic
• ready to use, easy to handle, pumpable liquid formulation
• stable to hard water, robust under process conditions
• water and energy conserving application process
• enhances whiteness index in case of full bleach process
• effectively removes chloramine after chlorine bleach process
• simple application process under high temp alkaline conditions
• develops alkaline reductive atmosphere in dye bath making it highly suited for material to be dyed with vat and sulphur dyes
• being inorganic does not increase cod and bod of effluent
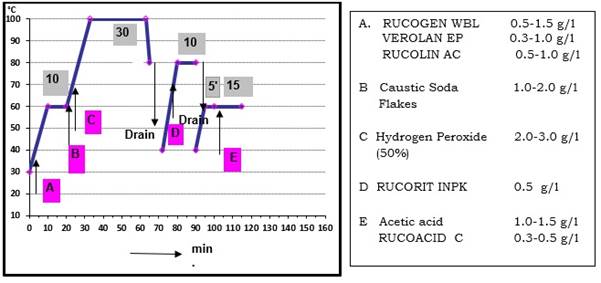
Given below is one representative process along with general recommended chemicals for bleaching of cotton knit material by exhaust application in soft flow machine, prepared for subsequent dyeing with Reactive dyes.
Given below is an example of effect of varying amount of residual bleaching agent on colour value of a medium black shade developed with a recipe of a combination of bi-functional reactive dyes.
 4. Enzymatic killing: This is the latest and advanced method of bleach clean-up specifically developed for neutralisation of residual hydrogen peroxide based oxidising agent in post bleaching process. Generally, the enzyme used here is ‘catalase’ which belongs to oxido-redutase class having classification no EC 1.11.1.6. This enzyme catalyses the decomposition of hydrogen peroxide to oxygen according to:
4. Enzymatic killing: This is the latest and advanced method of bleach clean-up specifically developed for neutralisation of residual hydrogen peroxide based oxidising agent in post bleaching process. Generally, the enzyme used here is ‘catalase’ which belongs to oxido-redutase class having classification no EC 1.11.1.6. This enzyme catalyses the decomposition of hydrogen peroxide to oxygen according to:
![]()
The enzyme is a bio catalyst produced by fermentation of microorganisms, acts very specifically on hydrogen peroxide and increases reaction speed by lowering the activating energy without any harmful side effects. Enzymes present a more convenient alternative because they are easier and quicker to use. With the increasingly important requirement for textile manufacturers to reduce pollution during production, the use of enzymes is rapidly gaining wider recognition because of their non-toxic and eco-friendly characteristics. The advantages of enzymatic treatment over other methods are:
• Complete and quick elimination of residual hydrogen peroxide
• Improved RFT and reproducibility in subsequent dyeing
• By-products are completely inert to dyestuffs and fabrics
• Suitable for combined peroxide killing and same bath dyeing
• Compatible with biopolishing process for combined application
• Reduced processing times, thus increasing production throughput
• Biodegradable – return to nature characteristic, no impact on pollution
The catalase enzyme has few limitations, in terms of its stability and applicability under specified narrow range of pH and temperature conditions. Generally, the conventional enzymes are susceptible to high temperature and strong pH conditions and get deactivated losing its effectiveness during bleach clean-up. Also, such conventional enzymes can be decomposed by enzyme poisons e.g. complexing agents, heavy metal ions.
RUCOLASE KAT is a specialty enzyme developed by RACL, formulated to withstand variation encountered during practical application conditions. The specific advantages include:
• easy to use, safe to handle liquid formulation for auto dispensing
• foam free characteristics that help improve machine runnability
• broad application spectrum from pH 4 to 9, maximum activity at pH 5.5-6.5
• stable up to 65oC, highly effective in range 45-55oC
• compatibility with selected complexing and/or neutralising agent
Given below is a schematic process flow of cotton yarn preparation regularly running at a leading textile unit in India.
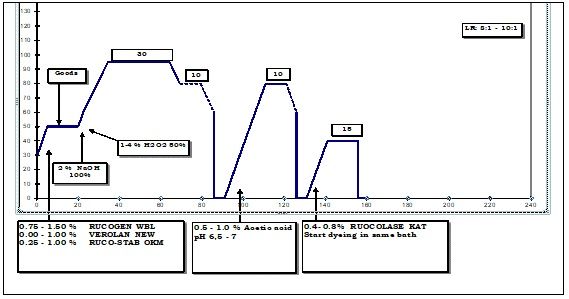
Assessment of Bleach Clean-up
To ensure effectiveness and efficiency of removal of residual bleaching agent, it is necessary to evaluate the same and various quantitative and qualitative methods of assessment are:
1. Titrimetric analysis – peroxide content in cotton is determined by titration of the extract in sulphuric acid with potassium permanganate solution. Violet colouration indicates presence of residual peroxide.
2. Chemical spot test – spotting textile substrate with titanyl chlroride indicator solution, yellowish-orange colour indicates presence of residual peroxide.
3. Dab strip test - this is the most commonly used and fairly accurate qualitative as well as quantitative method for assessing presence of residual peroxide after bleaching. Generally, Merck peroxide indicator paper strip is gently touched/dabbed on the textile substrate and the development and extent of bluish colour is compared with the indicator strip.





20240924091633.png)







Comments